Introduction
Knowing the energy amount that is left in a battery at use as compared to when the battery was fully charged informs the user how further the battery will keep performing before initiating the process of recharging. This process is known as the short-term measure of the battery capacity. The long-term measure of battery capability is known as the State of Health, which will be discussed later in another article. The State of Charge is defined as the measure of available capacity, which is expressed as the percentage of a certain reference of the battery's current capacity. The most preferred reference is the new battery-rated capacity, which is chosen because the capacity of the battery degrades as it gets old. This article will introduce the accuracy requirements of SOC and the methods used to determine SOCs.
The Accuracy Requirement of SOC
Lithium-ion batteries are the most chemically reactive cells. These cells require advanced BMS to keep them operating within the designed safe operating window and to ensure that they get the expected cycle life. Therefore, SOC knowledge is very important in the area of large-energy Lithium batteries. SOC is also very important in the Lithium batteries used in the automotive industry for efficient management of the flow of energy.
The SOC is used to determine the range of electric vehicles and when to switch on or off the engine in HEVs.
State of Charge Determining Methods
Several methods exist to determine the battery state of charge (SOC). Some of these methods are limited to certain cell chemistries, while most of them depend on measured cell parameters, which vary depending on the SOC. Some of these methods include:
- Direct SOC Measurement
- Specific Gravity Measurement
- Voltage-based SOC Estimation
- Current-based SOC Estimation
- Internal Impedance Measurement SOC Estimation
Direct SOC Measurement
This becomes very easy if the battery is measured at a constant rate of discharge. The charge stored in the battery is a product of current and the time that the current flows into the battery during charging. The disadvantage of this method is that, in practice, the amount of discharge current cannot be constant but gets diminished with battery discharge in a non-linear form. Therefore, the device used in this measurement must have the ability to integrate the current over discharge time. The second disadvantage is that the user needs to discharge the battery completely to know the charge stored in it. In most applications, the user should be able to know how much charge the battery has even without carrying out a discharge.
Specific Gravity Measurement
This method has been used as a custom in the determination of the lead-acid battery charge condition. It is made possible by determining the active chemical weight change. As the battery keeps discharging, the Sulphuric acid, which is the active electrolyte in this case, gets consumed, and its concentration in the battery water gets reduced. This ends up reducing the solution-specific gravity in direct proportionality to SOC. Hence, battery-specific gravity is another characteristic that can be used to indicate the battery's state of charge. Traditionally, a suction hydrometer has been used to carry out SG measurement but it is an inconvenient way of doing so. Modern-day technology has come up with electronic sensors that can give SG digital measurements. These sensors are connected directly to the battery cells and deliver the SG readings continuously. This method is not advisable for other types of battery cell chemistries.
Voltage-based SOC Estimation
The method makes use of each battery cell as the datum for SOC calculation. The outcome varies with the temperature, actual voltage, cell age, and discharge rates, and these factors compensation should be available for a reasonably accurate result. Check the graph in Figure 1 below to see the open circuit voltage and capacity at a constant discharge rate and temperature for the lead acid cell of high capacity.
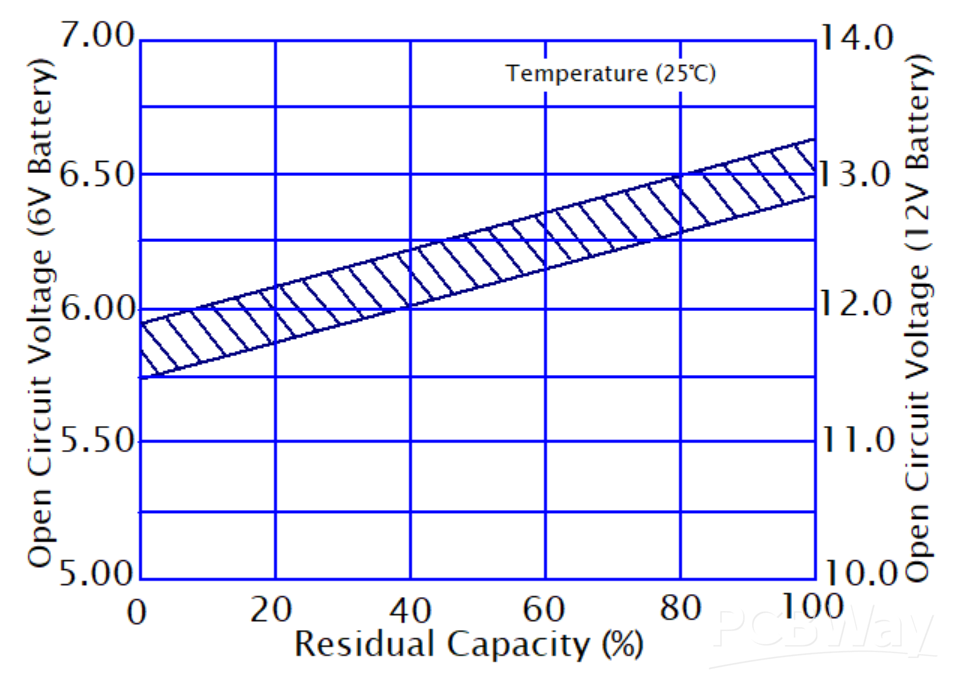
Figure 1: Open Circuit Voltage vs Residential Capacity at a Temperature of 250C Courtesy of Simon Mugo
This method is not suitable for some other cell chemistries like lithium-ion, which has a negligible change in its voltage throughout most of its charge/discharge cycle. Check the graph in Figure 2 for the Lithium-ion discharge process. There is a rapid fall of the cell voltage at the discharge end. The lithium-ion battery is not fully discharged because doing so will shorten its lifespan.
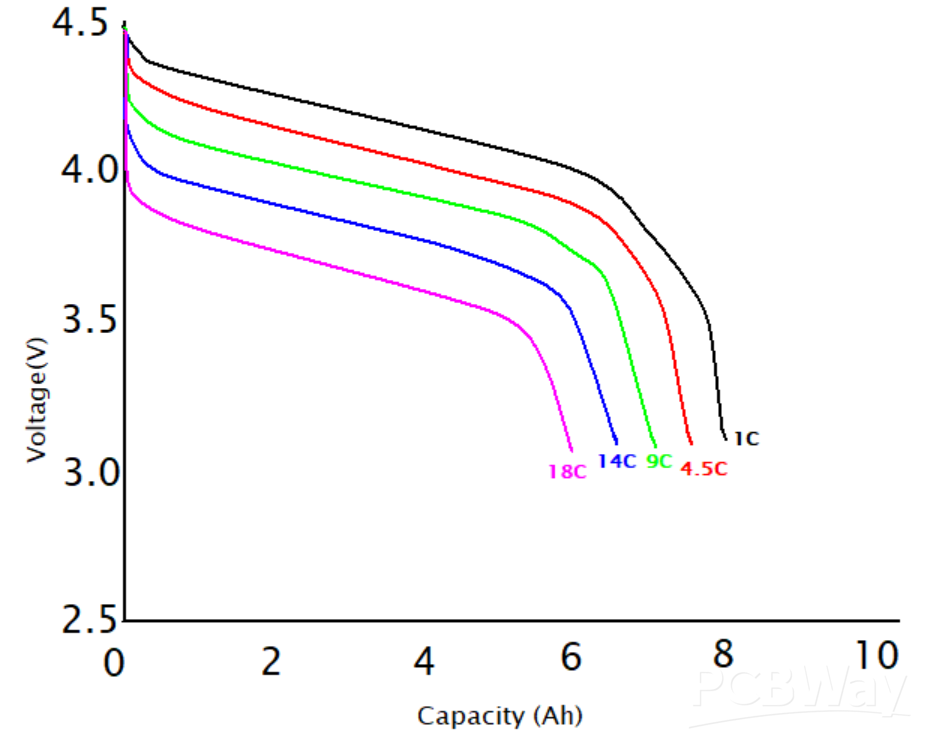
Figure 2: Battery Discharge Curves Courtesy of Simon Mugo
Current-based SOC Estimation
This method is also referred to as Coulomb counting. The energy present in any electric charge is equal to the integration of current over time that the charge is delivered, and the unit of measurement is the Coulomb. The capacity of energy in a cell can be estimated by measuring the current that enters or leaves the cell and then integrating it over the time it takes. This process is made successful by using a fully charged cell as the calibration reference. The method offers the best accuracy as compared to other testing methods since it involves direct measurement of the charge flowing in a cell.
The method is achieved through the use of three methods for testing currents, namely:
- · Current shunt: This involves measuring the voltage drop across high-precision, low-ohmic-value, sense, and series resistors connected between the load and the battery. The method is very inaccurate when low currents are involved and cause a small loss of power.
- · GMR: This is a magneto-resistive sensor that is a bit expensive but offers higher sensitivity with a very high signal level.
- · Hall-effect sensors and transducers: They are somehow expensive, but they are affected by noise and high currents.
This method of coulomb sensing depends on the electrical power that flows out of the cell without accounting for the self-discharge battery currents.
Internal Impedance Measurement SOC Estimation
During the battery's charging-discharging process, the active chemical competition changes as the chemical composition of the electrolyte is switched between the charged and discharged forms. The whole change is reflected in the changes in the battery cell impedance. Therefore, measuring the battery cell’s internal impedance is another way to decide the battery’s SOC. This method is not preferred because measuring impedance is a very difficult process in active cells, and interpreting measured data is also not that easy.
Summary
- · The state of charge (SOC) is defined as the measure of the battery’s available capacity, expressed as a percentage of a certain reference battery capacity.
- · The SOC's accuracy requirement is determined by the manufacturer's range, which includes voltage, current, and energy ratings.
- · Several methods are used in determining the SOC in batteries: direct and specific gravity measurement and voltage-based, current-based, and internal impedance measurement SOC estimation.
- · Direct SOC measurement involves measuring the battery’s constant rate of discharge, where the charge stored in the battery is a product of current and time of current flow.
- · Specific gravity measurement is best used in lead-acid batteries, where the SG is measured using a device known as a suction hydrometer.
- · Voltage-based SOC estimation uses the battery cell voltage to estimate the battery's state of charge.
- · Current-based SOC estimation, also known as coulomb counting, involves measuring the current flowing in and out of the cell to estimate the SOC.
- · The current-based SOC estimation involves three methods, namely current shunt, GMR, and hall-effect sensors.
- · Internal impedance measurement SOC estimation depends on the determination of the battery cell impedance.
 DIY Electronics
DIY Electronics
 Arduino
Arduino
 Hardware
Hardware
 Audio
Audio
 Computers & USB
Computers & USB
 Breakout Board Projects
Breakout Board Projects
 Home Automation
Home Automation
 LED Displays & Matrices
LED Displays & Matrices
 IoT
IoT
 Robotics
Robotics




